|
The individual classes take into consideration a product’s intended use, indications for use, and the level of risk associated with the product. The FDA recognizes three separate classes of medical devices: Classes I, II, and III. What problem will it solve? What assistance or therapy will your planned device deliver as a point of value to the end user? How will it work? The answers to these questions will determine the way the FDA will classify and therefore regulate your device. To do that, you need to be crystal clear about what your device is intended to do. Does My Product Need to Be Developed Under Design Controls?įirst things first: You need to determine whether or not Design Controls are required for your planned medical device. In this article, we’ll explore the basics of Design Controls, including whether or not they apply to your situation and an overview of how they are typically incorporated into product development. This process includes many layers of required documentation that show the FDA exactly how you have provided for the safety and efficacy of your new device. The first step in meeting the FDA’s requirements is to develop your medical device under Design Controls.ĭesign Controls, which are mandated by the FDA, represent a formalized approach to the development of Class II and Class III medical devices. 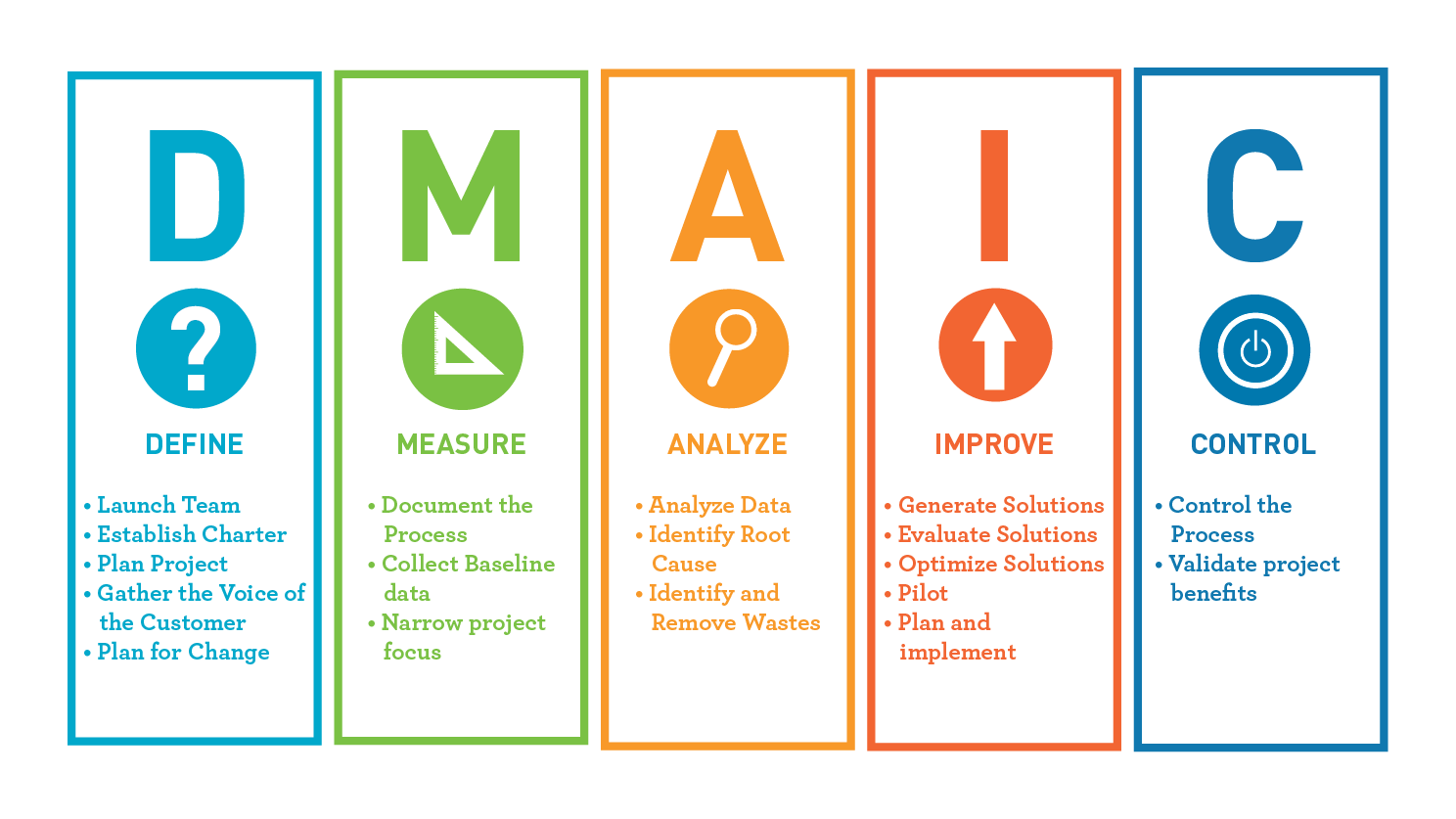
As the product manufacturer, it’s your job to ensure your device is both effective and safe for users whose health may be on the line.įor all but the lowest-risk medical devices, the Food and Drug Administration requires proof that you have done just that. Who wouldn’t be excited about that?īut medical devices also carry varying levels of risk that must be carefully managed. New medical devices offer fresh opportunities for your company to improve health outcomes, push the boundaries of medical innovation, and grow your business.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
